Learning TARGETS

*Identify the particles that make up atoms and describe their charges
*Identify and describe examples of different states of matter
*Describe and illustrate changes in state, in terms of arrangement and
motion of the atoms or molecules
*Diagram and describe the processes involved when a substance changes from state to state
*Explain how mass is conserved as it changes from state to state
----------------------------------------------------------------------------------------------------------------------
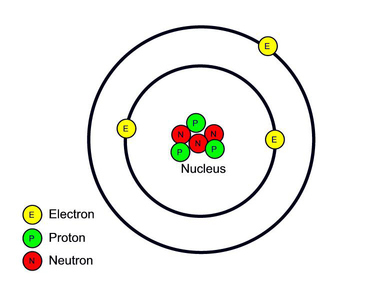
Atoms are particles that make up matter. Within an atom, you will find protons (postitive charge), nuetron (no charge), and electrons (negative charge).
*Identify and describe examples of different states of matter
*Describe and illustrate changes in state, in terms of arrangement and
motion of the atoms or molecules
*Diagram and describe the processes involved when a substance changes from state to state
*Explain how mass is conserved as it changes from state to state
----------------------------------------------------------------------------------------------------------------------
Atoms are particles that make up matter. Within an atom, you will find protons (postitive charge), nuetron (no charge), and electrons (negative charge).
--------------------------------------------------------------------------------------------------------
Memorize this short rhyme and it will help you understand our concepts:
I am matter.
I take up space.
Gravitational pull keeps me in place.
I have mass!
Matter is anything that takes up space and has mass
The motion of molecules or atoms will increase when temperature increases
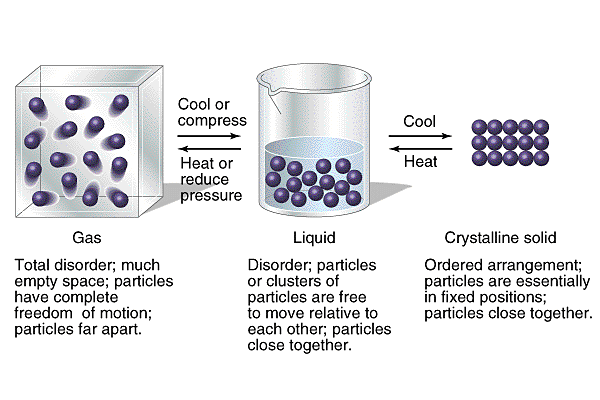
The three primary states of matter are explained below:
Memorize this short rhyme and it will help you understand our concepts:
I am matter.
I take up space.
Gravitational pull keeps me in place.
I have mass!
Matter is anything that takes up space and has mass
The motion of molecules or atoms will increase when temperature increases
The three primary states of matter are explained below:
Gas* No definite shape
* No definite volume *particles moving very FAST (sporadic movement) |
Liquid* No definite shape
* Definite volume *particles moving quickly (not as fast as within gas though) (sliding past each other) |
Solid*Definite shape
*Definite volume *particles moving slowly (vibrating) |
Check out these websites for a good visual of the motion of atoms within matter:
Test out your knowledge with this website (it has information and practice quizzes):
http://www.chem4kids.com/files/matter_states.html
View the prezi below to learn about the three phases of matter --
http://prezi.com/o_mg7rpdaxio/copy-of-three-states-of-matter/
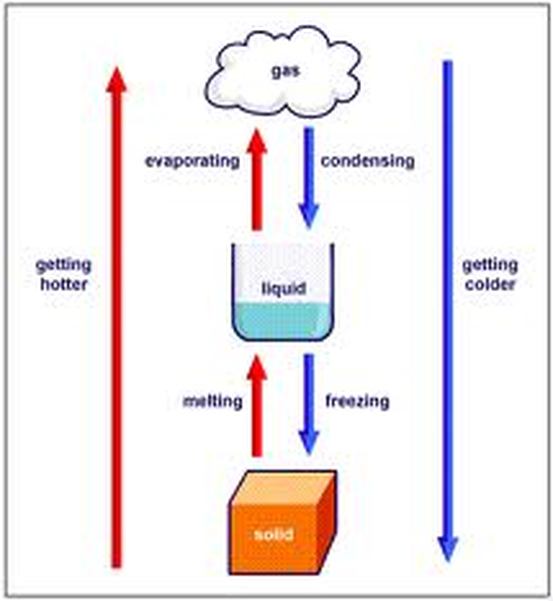
THERMAL ENERGY changes the state matter...here are the processes that are involved:
Liquid changes to solid = FREEZING (ex: water turns to ice in the freezer)
Liquid changes to gas = EVAPORATION (ex: puddle dries up on a hot day)
Solid changes to liquid = MELTING (ex: ice cream drips in the hot sun)
Solid changes to gas = SUBLIMATION (ex: dry ice turns to gas)
Gas changes to liquid = CONDENSATION (ex: mirror steams up during hot shower)
Gas changes to solid = DEPOSITION (ex: water vapor turns to ice on car windshield)
Liquid changes to gas = EVAPORATION (ex: puddle dries up on a hot day)
Solid changes to liquid = MELTING (ex: ice cream drips in the hot sun)
Solid changes to gas = SUBLIMATION (ex: dry ice turns to gas)
Gas changes to liquid = CONDENSATION (ex: mirror steams up during hot shower)
Gas changes to solid = DEPOSITION (ex: water vapor turns to ice on car windshield)
----------------------------------------------------------------------------
Within a CLOSED SYSTEM (sealed/isolated), the MASS of an substance will always remain constant, even when the substance changes from state to state, For example, if you place a chunk of ice with the mass of 30.6 grams in a closed ziplock bag and let it melt, the mass of water would still equal 30.6 grams.